  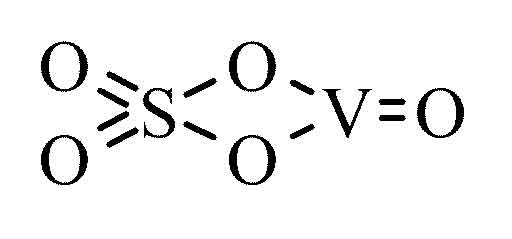
These sulfido rings are strongly oxidizing. When I dissolved CrCl 3 6H 2 O which is more properly represented by the formula CrCl 2 (H 2 O) 4 Cl (H 2 O) 2 in water, it slowly reacted with the solvent to give the violet-coloured Cr. +6 oxidation state) in the form of sodium. In potassium sulfate, K2SO4, the oxidation numbers are. In the environment, Cr(VI) is present in anionic form and is relatively mobile. oxidation number of each chromium and write the following equation: 2x + 7(-2). #S_8+S^(2-) rarr S_9^(2-)# where the sulfido anion is present as some form of ring.the formal oxidation state of #S# is #-2/9# of course, FORMALLY we got #8xxstackrel(0)S+stackrel(-II)S# OR #2xxstackrel(-I)S+6xxstackrel(0)S#. What is the oxidation number of chromium in dichromate ion, Cr2O72- 6+ In the sulfite ion, SO32-, what is the oxidation number of sulfur 4+ In the ammonium ion, NH4+, what is the oxidation number of hydrogen 1+ What is the oxidation number of hydrogen in H2 0 In the ion MnO4-, the oxidation number of manganese is. The production of basic chrome sulfate is based on the chemical reaction in which hexavalent chromium (ie. oxidation states of chromium show great contrast in their chemical behavior. So if you have got a slurry of sulfur in water.you can add some sulfide salt, and get the sulfur up into solution. In ZnCl2, the oxidation number of chlorine as Cl- is -1 and that of zinc as Zn2+ is +2. the yellow powder, that we can find in a bottle, is mighty insoluble stuff.and this is probably the #S_8# molecule, i.e. The oxidation number of a monatomic ion is the charge on that ion. Now it is a FACT that sulfur flowers, i.e. (And note that both sulfur and oxygen are Group 16 chalcogens, and they should sometimes have the same oxidation number!)Īnd so FORMALLY we gots #stackrel(-II)S#, AND #stackrel(+VI)S# and we may continue the formalism, and assign an AVERAGE oxidation number of #(-II+VI)/2=+II# for both sulfurs.Īnd another way of looking at this is to consider the #S^(2-)# ion.which, I hope you will agree has a formal #-II# oxidation state. And with this formalism, we gots #stackrel(-II)S+stackrel(+VI)S+3xxstackrel(-II)O=-2#, the charge on the ion as required. The oxidation number of the sulfur atom in the SO42-ion must be +6, for example, because the sum of the oxidation numbers of the atoms in this ion must equal -2. to speciate as little as 50 ppm of chromium. 
#4xx-II+VI=-2#, if you will forgive me mixing Roman and Arabic numerals (of course you will).Īs regards #"thiosulfate ion"#, #S_2O_3^(2-)#, I have always liked to think that one of the oxygens has been replaced by a #"sulfide ion"# with PRECISELY the same oxidation state as the oxygen, i.e. relative heights of the pre-edge peaks for the different Cr oxidation states in XANES spectra, can be used. Salts of chromium (II) have a bluish color. The compounds are colored chromium (II) oxide is black, and the hydroxide is yellow. Sodium Dichromate Na2Cr2O7 or Cr2Na2O8 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological. For the purposes of redox we can consider that it has an ionic charge of +6. Chromium in the oxidation state of +2 Chromium (II) oxide and hydroxide CrO and Cr (OH) are compounds which display typical base properties. As always, the weighted sum of the oxidation numbers is equal to the charge on the ion, i.e. For example, in sulfuric acid, the sulfur is in the VI (+6) oxidation state. We assign oxygen an oxidation number of 2 and write the equation In SO42, the polyatomic anion has a charge of 2. In SO32, the polyatomic anion has a charge of 2. In S2, the oxidation number of sulfur is 2. #SO_4^(2-)# we have #"sulfur(+VI)"# and #"oxygen(-II)"#. Solution We can begin by recalling that the charge on an ion corresponds to the sum of the oxidation numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
